
2026-01-14
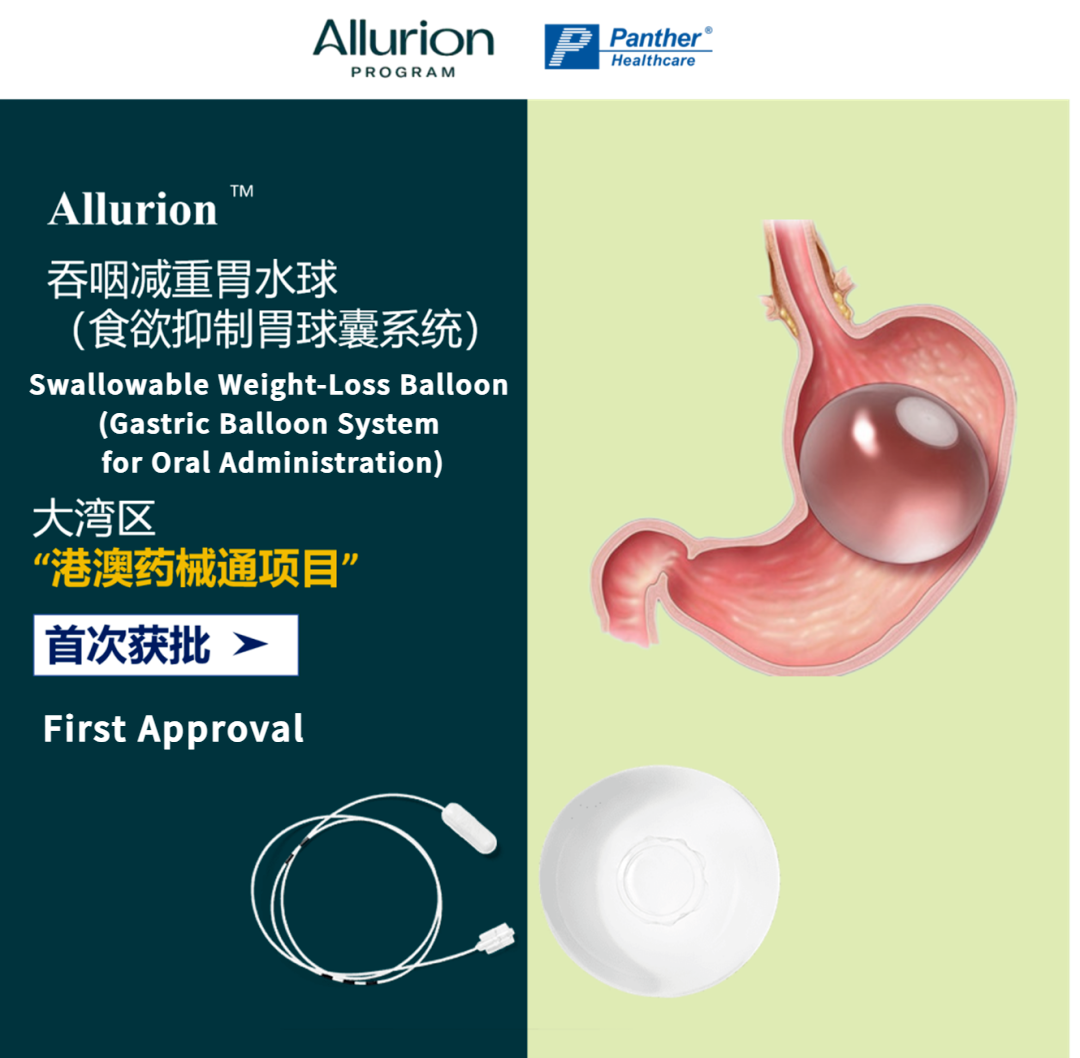
An international, cutting-edge swallowable gastric balloon for weight loss has officially received approval from the Guangdong Provincial Medical Products Administration. Leveraging the Hong Kong and Macao Medicine and Equipment Connect Policy, the product has been introduced at Foshan Fosun Chancheng Hospital, ushering in a new era of non-invasive weight management.
1. Policy Implementation: International Innovative Weight-Loss Device Enters Greater Bay Area Clinical Practice
The newly approved Allurion Appetite Suppression Gastric Balloon System is the world’s first gastric balloon that requires no endoscopy, no anesthesia, and can be placed through natural swallowing.
As a tangible outcome of the Hong Kong and Macao Medicine and Equipment Policy, the system has successfully entered clinical use at Foshan Fosun Chancheng Hospital. It offers overweight and obese patients in the Greater Bay Area access to a globally synchronized, non-surgical weight-loss solution, while also introducing a novel clinical option to China’s non-surgical, device-based weight management landscape.
2. Core Advantages: Redefining the Non-Invasive Weight-Loss Experience
Compared with traditional intragastric balloons, the Allurion swallowable gastric balloon significantly enhances patient experience through technological innovation. Its key advantages include:
Eligible population: Suitable for overweight and obese patients with a Body Mass Index (BMI) ≥ 27 kg/m².
15-minute outpatient procedure:
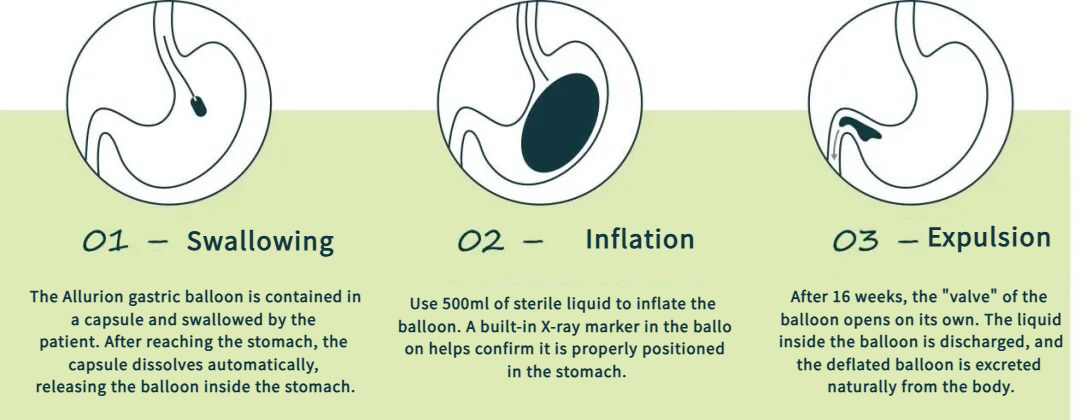
The balloon is preloaded inside a capsule. Patients simply swallow it with water, and placement is completed in approximately 15 minutes in an outpatient setting, with no hospitalization required.
Fully non-invasive management: No gastroscopic placement or removal is needed, avoiding risks associated with endoscopic procedures. After remaining in the body for approximately 16 weeks, a release valve opens automatically, allowing the saline to be discharged; the balloon material is then naturally excreted through the gastrointestinal tract.
Controllable clinical safety profile: Clinical data indicate a low overall incidence of adverse events.
3. Clinical Value: Multidimensional Health Improvement

The Allurion (Anruiren) swallowable gastric balloon has been launched in more than 60 countries and regions worldwide, with cumulative sales exceeding 200,000 units. Its clinical value has been extensively validated in real-world practice.
4. Multidisciplinary Collaboration: Upgrading Comprehensive Weight-Management Strategies
As the prevalence of overweight and obesity continues to rise in China, patient demand for safe and effective weight-loss interventions is becoming increasingly urgent. In response, Foshan Fosun Chancheng Hospital has established a multidisciplinary team (MDT) and developed a full-cycle health management system incorporating nutritional counseling and behavioral intervention, providing patients with structured, evidence-based weight management support.
The introduction of the swallowable gastric balloon under the Hong Kong and Macao Medicine and Equipment Policy adds an internationally advanced treatment option to the hospital’s portfolio. This further enriches the diversity of weight-loss solutions available, enabling more precise and personalized treatment pathways tailored to individual patient needs.
Important Notice⚠️
This product is a medical device and must be used under the guidance of healthcare professionals. Contraindications and precautions are detailed in the product instructions for use.
Source From: Foshan Fosun Chancheng Hospital Official Subscription Account

