
2025-12-25
For patients with severe illnesses, “prohibitive costs” and “lack of access to cutting-edge therapies” are often daunting challenges. With the continuous optimization of the Foshan Medical Insurance policy, a landmark change arrives in 2026: for the first time, drugs and medical devices approved under the “Greater Bay Area (GBA) Drug and Device Access” policy are included in the reimbursement catalog. Insured patients receiving these treatments at designated hospitals can now claim 50% reimbursement for cumulative annual expenses exceeding 40,000 RMB, with an annual cap of 200,000 RMB.
Foshan Fosun Chancheng Hospital (“Foshan Chancheng Hospital”), a subsidiary of Fosun Health and the first hospital in Foshan designated under the GBA Access policy, has already introduced over 30 international innovative drugs and devices. The four treatments now covered by insurance are among its most clinically established and urgently needed by patients, targeting three critical areas: osteoporosis, breast cancer, and coronary artery disease.
Four Innovative Treatments: Precision Solutions for Clinical Challenges
01 Romosozumab: Fortifying Bone Defense for High-Risk Patients
Osteoporosis, known as a "silent killer," poses a particularly high fracture risk for postmenopausal women. Patients with recent fragility fractures, those who fracture despite conventional therapy, or those with a bone mineral density T-score < -3.0 often face limited treatment options.
Romosozumab offers a new direction for postmenopausal women with severe osteoporosis at high fracture risk. With its unique mechanism of action modulating bone metabolism, it helps improve bone mass and microarchitecture to reduce the risk of fragility fractures.
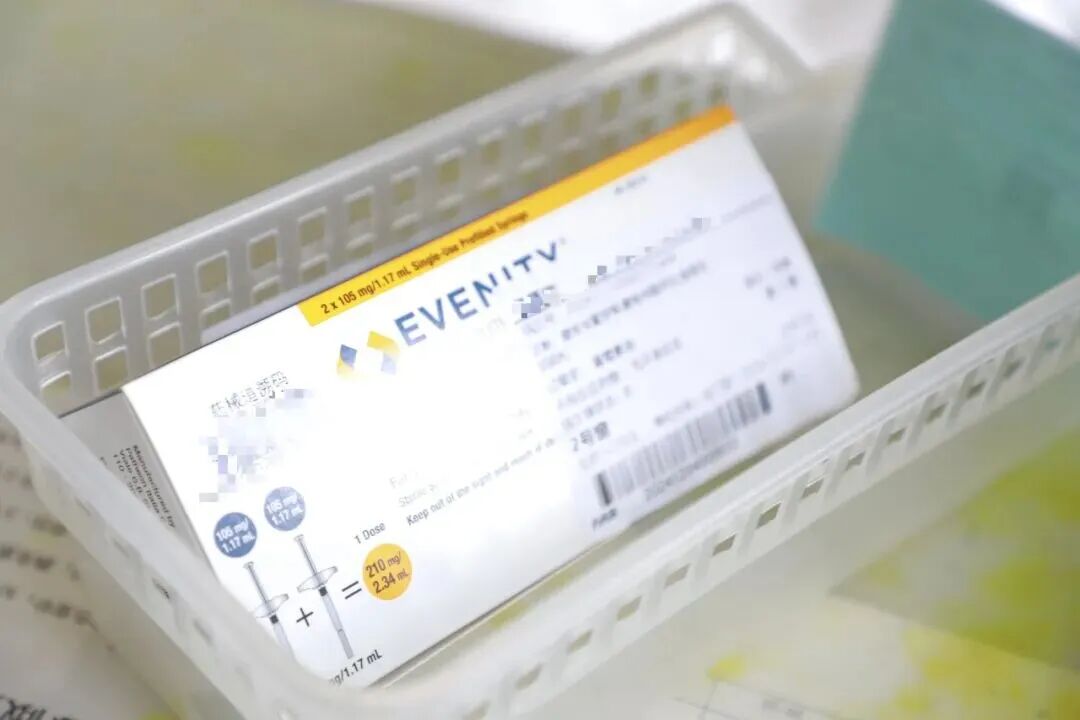
Romosozumab
02 Elacestrant (Oral SERD): A New Oral Hope for Breast Cancer Patients
Hormone receptor-positive (HR+) breast cancer accounts for about 70% of cases. Patients with advanced disease often confront endocrine therapy resistance, especially those with ESR1 mutations, leaving them with very limited subsequent options.
Elacestrant, the world's first approved oral Selective Estrogen Receptor Degrader (SERD), precisely targets ESR1-mutated, resistant cancers. It effectively inhibits mutant estrogen receptor α activity and degrades the receptor, significantly extending progression-free survival. Compared to traditional injectable therapies, the oral formulation greatly enhances long-term treatment convenience and patient adherence, while demonstrating a favorable safety profile in clinical trials.

Elacestrant (Oral SERD)
03 Diamondback 360 Coronary Orbital Atherectomy System: A Powerful Tool to Tackle Coronary Calcification
"Nothing is more challenging than severe coronary calcification" is a consensus among cardiologists. Heavily calcified lesions can prevent interventional devices from passing through, drastically increasing procedural difficulty and risk, often limiting traditional treatment approaches.
Abbott's Diamondback 360 system tackles this with a high-speed rotating crown that precisely ablates superficial calcium while its “orbital atherectomy” mechanism fractures deeper calcium, clearing the path for stent implantation and significantly improving success rates in complex cases. Approved by the U.S. FDA and the EU, it is indicated for patients with severe de novo coronary artery calcification amenable to percutaneous intervention.

Abbott's Diamondback 360 system
04 Freesolve Bioresorbable Magnesium Alloy Scaffold: The 'Invisible Guardian' for CAD Patients
Traditional metallic stents remain in the artery permanently, which may increase long-term risks of thrombosis and restenosis, a significant concern especially for younger patients. Polymer-based bioresorbable scaffolds, meanwhile, have faced challenges with insufficient radial strength and long degradation times.
The Freesolve Sirolimus-Eluting Coronary Bioresorbable Magnesium Alloy Scaffold System, the world's first magnesium alloy bioresorbable coronary scaffold, degrades and is absorbed by the body within one year, leaving no permanent metallic implant and allowing better restoration of the vessel's natural elasticity. Compared to polymer scaffolds, it offers superior radial strength with thinner struts, potentially reducing risks of acute and late complications. Three-year follow-up data shows a target lesion failure rate of just 3.5%.
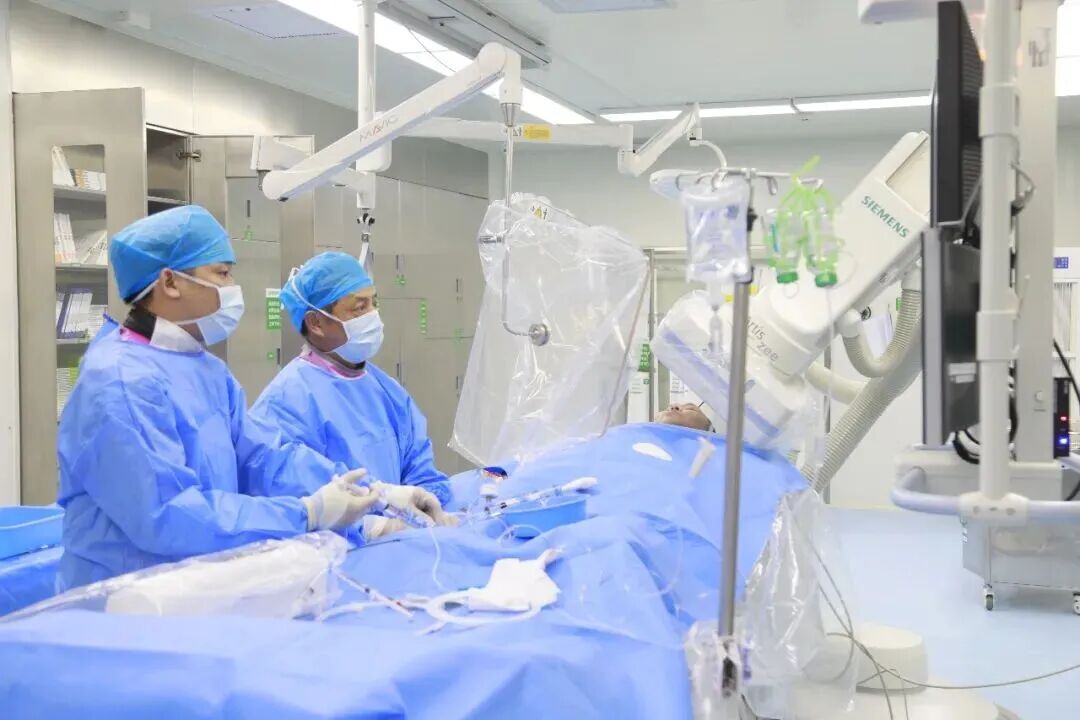
Stent Implantation
As Foshan's first GBA Access designated institution, Foshan Chancheng Hospital is committed to advancing integrated, high-quality healthcare in the Greater Bay Area. By leveraging this policy, it actively introduces global innovations, shortening the "time lag" for mainland patients to access advanced treatments.
The inclusion of these four innovative therapies in the Foshan insurance plan represents a powerful synergy of pro-patient policy and medical innovation. Moving forward, Foshan Chancheng Hospital will continue to leverage its multi-disciplinary strengths to deepen the clinical application of such innovations, providing patients with higher-quality, more convenient, and accessible medical care—empowering more people to receive world-class treatments right at their doorstep.

